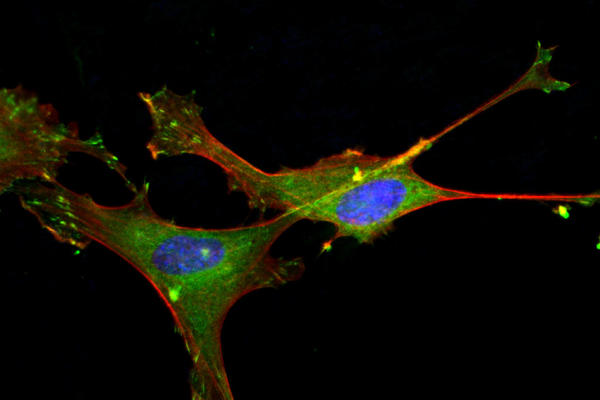
Treatment of congenital or other acquired diseases has prompted the development of bioactive materials with multifunctional attributes with appropriate mechanical properties. Assessing the physico-mechanical properties of these novel materials is as important as determining the interaction they have with the cellular environment. These assessments include determining their biocompatibility either via direct or indirect testing (following ISO recommendations) against cell types that will be directly interacting with the material. This includes measuring cell metabolic activity post exposure using colorimetric assay and/or determining cell morphology via immunofluorescence. For materials designed to support bone growth, the materials can be tested for their ex-vivo bioactivity using modified simulated body fluid to determine apatite precipitation (XRD for crystal structure, SEM for apatite structure and EDS for chemical verification). The in vitro bone forming capability can be tested using osteoprogenitor cells and the osteogenic differentiation can be assessed using early and late markers of differentiation via ELISAs or immunofluorescence. Additionally, we also perform antibacterial assessments against single or multi-species in static or bioreactor setting dependent on the material design and application criteria.